Summary
Soil chromatography (Pfeiffer Chroma-Test), is a qualitative method used in biodynamic agriculture to assess soil, biofertilizers and crops quality/health. For the workshop we took soil samples around the Tsinghua University Campus and we analyze them with this method. During the workshop we covered the basics of the soil chromatography method and afterword we discussed on how to get information from the chromas. We ended up the workshop outlining the feasibility to apply machine learning on the chromas to correlate with standard lab soil tests (ie. carbon content, nutrients).
Notes
Convener/Facilitator: Nano/Tamara/Rachel
Participants: Greg, Andryi, Saad, Oliver and almost all the temporary inhabitants of Open Fiesta Lab.
Special thanks to @leandro.mastrantonio who show me this method and give all material to do the workshop
Small resume of the process based on RESTREPO RIVERA, J. R; PINHEIRO, S. Cromatografía – Imágenes de vida y destrucción del suelo. Juquira Candiru Satyagraha, 2011. (Picture by Rachel)
Paper filter preparation with Silver Nitrate (AgNO3)
Filter paper is the stationary medium in this chromatographic method. Whatman N°1 or N°4 are the ones recommended but similar paper filters can be used. In our case we used a chinese brand of 15cm diámeter.
Silver nitrate is the substance that “develops” (in the presence of indirect sunlight) different patterns and colors when reacts with the different components extracted from soil.
To make a 0.5% AgNO3 solution we used a 0.5 g of pure silver nitrate salt in 100 ml of pure water.
ATTENTION!! This part of the process requires as low light as you can get!!
Procedure: In order to embed the paper filter with the solution we made some paper filter squares of 2 cm x 2 cm to make a wick. Then the wick is placed on a small hole pierced on the center of the paper filter. Then, we poor the solution in a petri dish and put the wick in contact with the solution (as in the picture). The solution embeds the paper by capillary action. Then we remove the wick and put the paper to dry in a closed box.
(not dark enough!!??) (Picture by Tamara)
Soil extraction and analysis
Ten grams of sieved (2 mm) soil of each sample were placed in a flask containing 50 ml of 1% aqueous solution of NaOH for extraction. NaOH is used to extract organic matter. The flask was agitated by hand at the beginning of the extraction, and after 15 min, 1, and 2 h. After the last agitation the flask was left for another 2 h for extraction and sedimentation. After a total of approximately 4 h the supernatant was collected and analyzed. We follow the same embedding procedure as for the AgNO3 with every soil sample. We made duplicates from all of them at the same time to check for repeatability.
Soil extraction with NaOH for aprox 6 hours (Picture by Rachel)
Results
(Not so) Blanks
We made several blank tests in order to understand better the results of this method. We made blanks for pure water (without NaOH), tap water (with NaOH) and for the 1% NaOH solution.
Soil samples
Interpretation basics
Taken from: Livia Bischof Pian, 2017, Chromatography of Pfeiffer: Principles, method and use in perception of soils, Annual project 2016/2017, Landbauschule Dottenfelderhof Fachschule für Biologisch-Dynamische Landwirtschaft. https://www.dottenfelderhof.de/fileadmin/images/landbauschule/projektarbeiten/Projektarbeit_Livia_Pian2016-17_ingles.pdf
“The minerals, organic molecules, proteins, vitamins and humic substances contained in the sample react with silver, forming complexes that result in different colours, forms and secondary structures such as arrows, zones and circles. For example, samples with higher concentration of nitrogen solubilize silver oxide, forming the Amin silver complex [Ag(NH3)2]+ which results in a white coloration in the central zone of chroma.
Besides the chromas being unique, representing unique forms and colours from samples, there are general characteristic patterns for each substance group. For example, soil chromas are similar to each other, with similar general characteristics, but very different from chroma of milk or compost. “
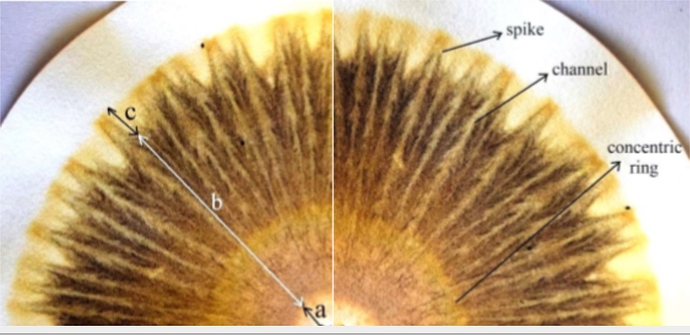
“The chroma interpretation involves the image description, to standardize this description we use names for the typical forms observed. First we separate the chroma into 3 zones: a) central zone, b) inner zone and c) outer zone. The forms found in the inner zone called channels and concentric rings and the form represent the channel that advances throw outer zone is called spike
For each zone and shape found in the chroma, general characteristics and functions of the soil can be related. By the central zone pass all the substances and are distributed in the chroma, the first reaction with the silver occur here and the formation or silver oxides is more intense. This zone is also called “aeration zone” because reflects the aeration conditions and the presence of nitrogen. little aeration and tendency to anaerobic metabolism would have dark zone, and aerobic soils present a yellow coloration and cream. In soils with a high presence of soluble nitrogen this zone assumes white coloration. The inner zone, called the ‘mineral and organic matter zone’, concentrates the reactions with minerals and humus of the soil. The concentric rings present in this zone indicate the presence of soluble minerals, if they are detached and without integration with other rings, may be indicative an excess of soluble minerals. Already when rings are integrated and the colours evolve continuously, it indicates that the minerals are in equilibrium and present in the form of humus-clay complexes. The humus accumulates at the end of this zone, giving brown colours, if the brown colour evolves from beginning to end of the zone in continuous way indicates that the humus is integrated with the minerals and available for the plants. If the brown coloration is disconnected from the more inner zone indicates that humus is immobilized. The outer zone is the expression of complex substances of high molecular weight, such as proteins, enzymes and vitamins (Pfeiffer, 1984). Substances created by metabolism of microorganisms present in soils, therefore indicate the biodiversity of the soil. This zone is very connected with the presence and distribution of canals and spikes. The number and the rhythmic form and distribution of channels represent the balance of soil microorganisms. In addition, the channels are a pattern of the integration between all zones. Soils with a balance and integration between mineral, humus and microorganisms fractions present channels that start in the central zone and go to the external zone.”
What we learned
The workshop was more of a DIWOrkshop (short residence??) because it was my first time running soil chromatographies and I shared the experimentation with others, specially Tamara and Rachel that teach me a lot about manual labour and procedures in the lab.
By just looking at dry soil it was possible to see a difference in color and texture. This difference was also evident during the extraction, the ones with darker colors were the more “organic”.
As a first approach to the method it was interesting to see that different soils show different patterns, and they are very beautiful. Also, the chromas from the same sample look very similar and tests performed with a few days of difference looks very similar too.
The general rule of the more complex the pattern the most organic the soil was consistent with the chromas. In the less organic soils (sand path, construction material) the concentric rings were not connected with channels and didn’t have a more rich outer zone as under fern or by the lake samples. However, to have a more profound read of the chromas we need to experiment a lot more with the method and have some background on other soil properties, processes and history.
We were concerned about why the blank 1% NaOH solution is not blank? Then we realize that this is ok and when the sample runs on this silver nitrate there is immediate formation of silver hydroxide (AgOH), a relatively unstable substance, which quickly forms a precipitate of silver oxide (Ag2O). This silver oxide reacts with light revealing dark coloration.
We also learned that we need to be very careful with the disposal of the solutions. For AgNo3 solution we need to add NaCl to neutralize it because Ag is poisonous.
There are several details that need to be addressed in order to standardize the protocol and make it more reproducible (ie, how many blanks, copies, lights to reveal, etc)
Follow up actions
After some discussions, previous and during the workshop, we believed that would be interesting to run a small batch of soil samples to see if it is possible to apply machine learning methods to the chroma images and try to correlate its colors and patterns with standard lab soil tests (ie. carbon content, nutrients).
To do this, Greg from Our-Sci, offered to run chromas on a group of the remaining soil samples of the Real Food Campaing already analyzed for its database and then apply AI to see what can be observed as a proof of concept..
This is not the first attempt to “scientifically validate” the soil chroma method [4, 6] and there are several cases were the method is used to differentiate between organic and traditional farming soil and crops [1, 2].
It is also interesting to consider that
“The validation of the method by academy allows the popularization and use by more farmers, what already happens nowadays in India.This motivation is valid and I will not question it. After all is an important evolution of the use and interpretation of chroma from a material point of view. But the relation of farmers with the chromatography continues to be through numbers. In the end an image becomes numbers, and all the potential that an image has is lost [3]”
Finally, I think is also important to consider the use of this charming “soil pictures” to support all the narrative of transition to more sustainable farming and soil management practices [5].
Bibliography
- Dietrich Knorr (1982) Use of a Circular Chromatographic Method for the Distinction of Collard Plants Grown under Different Fertilizing Conditions, Biological Agriculture & Horticulture: An International Journal for Sustainable Production Systems, 1:1, 29-38.
- Maria Olga Kokornaczyk, Fabio Primavera, Roberto Luneia, Stephan Baumgartner & Lucietta Betti (2016): Analysis of soils by means of Pfeiffer’s circular chromatography test and comparison to chemical analysis results, Biological Agriculture & Horticulture.
- Livia Bischof Pian, 2017, Chromatography of Pfeiffer: Principles, method and use in perception of soils, Annual project 2016/2017, Landbauschule Dottenfelderhof Fachschule für Biologisch-Dynamische Landwirtschaft.
- Masedo, M. T., 2016, Desarrollo y Caracterización del método de Dinamólisis Capilar para el Análisis de Suelos Contaminados, Estudios de los suelos de Linares. Madrid.
- Restrepo Rivera, J. R; Pinheiro, S. Cromatografía – Imágenes de vida y destrucción del suelo. Juquira Candiru Satyagraha, 2011.
- Khemani D, Joseph MM, Variganti S. 2008. Case based interpretation of soil chromatograms. In: Althoff HD, Bergmann R, editors. Advances in case based reasoning. Proceedings of the 9th European conference in case based reasoning; 2008 Sep 1–4; Trier, Germany; p. 587–599.
Resources
You can find most of the info cited here on this folder
Including the book Cromatografía de suelos - Jairo Restrepo (in spanish)
Links
http://wiki.artscienceblr.org/wiki/index.php/Circular_chromatography
http://lamierdadelavaca.com/
List of materials for the workshop
Reagents:
Pure Silver Nitrate Salts(analytical grade). 10 g
Sodium hydroxide (analytical grade). 50 g
Distilled water around 2-3 liters
Materials:
Scale 0.5 g precision.
Filter papers 15cm diameter. Whatman Nº4 or Sartorious 1288
1 Graduated cylinder 100 ml
10 Disposable Petri dishes 5 cm diameter
10 Disposable Petri dishes 10 cm diameter
6 Erlenmeyer flasks 125 ml
1 amber flask 100 ml
1 liter cheap beaker
1 chep small mortar
Paper tissue
5 ml syringe with needle
2mm sieve
Related links
https://www.tandfonline.com/doi/abs/10.1080/01448765.2010.10510429